Any sufficiently convoluted explanation for biological phenomena is indistinguishable from epigenetics.
Epigenetics is everywhere. Nary a day goes by without some news story or press release telling us something it explains.
Why does autism run in families? Epigenetics.
Why do you have trouble losing weight? Epigenetics.
Why are vaccines dangerous? Epigenetics.
Why is cancer so hard to fight? Epigenetics.
Why a cure for cancer is around the corner? Epigenetics.
Why your parenting choices might affect your great-grandchildren? Epigenetics.
Epigenetics is used as shorthand in the popular press for any of a loosely connected set of phenomenon purported to result in experience being imprinted in DNA and transmitted across time and generations. Its place in our lexicon has grown as biochemical discoveries have given ideas of extra-genetic inheritance an air of molecular plausibility.
Biologists now invoke epigenetics to explain all manner of observations that lie outside their current ken. Epigenetics pops up frequently among non-scientists in all manner of discussions about heredity. And all manner of crackpots slap “epigenetics” on their fringy ideas to give them a veneer of credibility. But epigenetics has achieved buzzword status far faster and to a far larger extent than current science justifies, earning the disdain of scientists (like me) who study how information is encoded, transferred and read out across cellular and organismal generations.
This simmering conflict came to a head last week around an article in The New Yorker, “Same but Different” by Siddhartha Mukherjee that juxtaposed a meditation on the differences between his mother and her identical twin with a discussion of the research of Rockefeller University’s David Allis on the biochemistry of DNA and the proteins that encapsulate it in cells, that he and others believe provides a second mechanism for the encoding and transmission of genetic information.
Although Mukherjee hedges throughout his piece, the clear implication of the story is that Allis’s work provides an explanation for differences that arise between genetically identical individuals, and even suggests that they open the door to legitimizing the long-discredited ideas of the 19th century naturalist Jean-Baptiste Lamarck who thought that organisms could pass beneficial traits acquired during their lifetimes on to their offspring.
The piece earned a sharp rebuke from many scientists, most notably Mark Ptashne who has long led the anti-epigenetics camp, and John Greally, who published a lengthy take-down of Mukherjee’s piece on the blog of evolutionary biologist Jerry Coyne.
The dispute centers on the process of gene regulation, wherein the levels of specific sets of genes are tuned to confer distinct properties on different sets of cells and tissues during development, and in response to internal and external stimuli. Gene regulation is central to the encoding of organismal form and function in DNA, as it allows different cells and even different individuals of a species to have identical DNA and yet manifest different phenotypes.
Ptashne has studied the molecular basis for gene regulation for fifty years. His and Greally’s critique of Mukherjee, or really Allis, is rather technical, and one could quibble about some of the specifics. But his main points are simple and difficult to refute:
- There is essentially no evidence to support the idea that chemical modification of DNA and/or its accompanying proteins is used to encode and transmit information over long periods of time.
- Rather than representing a separate system for storing and conveying information, a wide range of experiments suggests that the primary role of the biochemistry in question is to execute gene expression programs encoded in DNA and read out by a diverse set of proteins known as transcription factors that bind to specific sequences in DNA and regulate the expression of nearby genes.
In one way this debate is incredibly important because it is ultimately about getting the science right. Mukherjee’s piece contained several inaccurate statements and, by focusing on one aspect of Allis’s work, gave an woefully incomplete picture of our current understanding of gene regulation.
Any system for conveying information about the genome – which is what Mukherjee is writing about – has to have some way to achieve genomic specificity so that the expression of genes can be tuned up or down in a non-random manner. Transcription factors, which bind on to specific DNA sequences, provide a link between the specific sequence of DNA and the cellular machines responsible for turning information in DNA into proteins and other biomolecules. Small RNAs, which can bind to complementary sequences in DNA, also have this capacity.
But there is scant evidence for sequence specificity in the activities of the proteins that modify DNA and the nucleosomes around which it is wrapped. Rather they get their specificity from transcription factors and small RNAs. That doesn’t render this biochemistry unimportant – the broad conservation of proteins involved in modifying histones shows they play important roles – but ascribing regulatory primacy to DNA methylation and histone modifications is not consistent with our current understanding of gene regulation.
Something is, however, getting lost in this back-and-forth , as one might come away with the impression that this is disagreement about whether cells and organisms can transmit information in a manner above and beyond DNA sequence. And this is unfortunate, because there really is no question about this. Ptashne and Allis/Mukherjee are arguing about the molecular details of how it happens and about how important different phenomena are.
Various forms of non-Mendelian information transfer are well established. The most important of which happens in every animal generation, as eggs contain not only DNA from the mother, but also a wide range of proteins, RNAs and small molecules that drive the earliest stages of embryonic development. The particular cocktail left by the mother can have profound effects on the new organism – so called “maternal effects”. These effects can be the result of both the mothers genotype, the environment in which she lives, and, in various ways, her experiences during her life. (Such phenomena are not limited to multicellular critters – single-celled organisms distribute many molecules asymmetrically when they divide, conferring different phenotypes to their different genetically identical offspring).
Many maternal effects have been studied in great detail, and in most cases the transmission of state involves the transmission of different concentrations and activities of proteins (including transcription factors) and RNAs. That is the transmitted DNA is identical, but the state of the machinery that reads out the DNA is different, resulting in different outcomes.
However there are some good examples in which modifications to DNA play an important role in the transmission of information across generations – most notably with “imprinting”, in which an organism preferentially utilizes the copy of a gene it got from one of its parents independent to the exclusion of the other in a way that appears to be independent of the sequence of the gene. Imprinting, which is a relatively rare, but sometimes important, phenomenon appears to arise from parent-specific methylation of DNA.
Could the histone modifications that Allis studies and Mukherjee focuses on also carry information across cell divisions and generations? Sure. Our understanding of gene regulation is still fairly primitive, and there is plenty of room for the discovery of important inheritance mechanisms involving histone modification. We have to keep an open mind. But the point the critics of Mukherjee are really making is that given what is known today about mechanisms of gene regulation, it is bizarre bordering on irresponsible to focus on a mechanism of inheritance that only might be real.
And Mukherjee is far from the only one to have fallen into this trap. Which brings me to what I think is the most interesting question here: why does this particular type of epigenetic inheritance involving an obscure biochemical process have such strong appeal? I think there are several things going on.
First, the idea of a “histone code” that supersedes the information in DNA exists (at least for now) in a kind of limbo: enough biochemical specificity to give it credibility and a ubiquity that makes is seem important, but sufficient mystery about what it actually is and how it might work that people can imbue it with whatever properties they want. And scientists and non-scientists alike have leapt into this molecular biological sweet spot, using this manifestation of the idea of epigenetics as a generic explanation for things they can’t understand, a reason to hope that things they want to be true might really be, and as a difficult to refute, almost quasi-religious, argument for the plausibility of almost any idea linked to heredity.
But there is also something more specifically appealing about this particular idea. I think it stems from the fact that epigenetics in general, and the idea of a “histone code” in particular, provide a strong counterforce to the rampant genetic determinism that has dominated the genomic age. People don’t like to think that everything about the way they are and will be is determined by their DNA, and the idea that there is some magic wrapper around DNA that can be shaped by experience to override what is written in the primary code is quite alluring.
Of course DNA is not destiny, and we don’t need to evoke etchings on DNA to get out of it. But I have a feeling it will take more than a few arch retorts from transcription factor extremists to erase epigenetics from the zeitgeist.
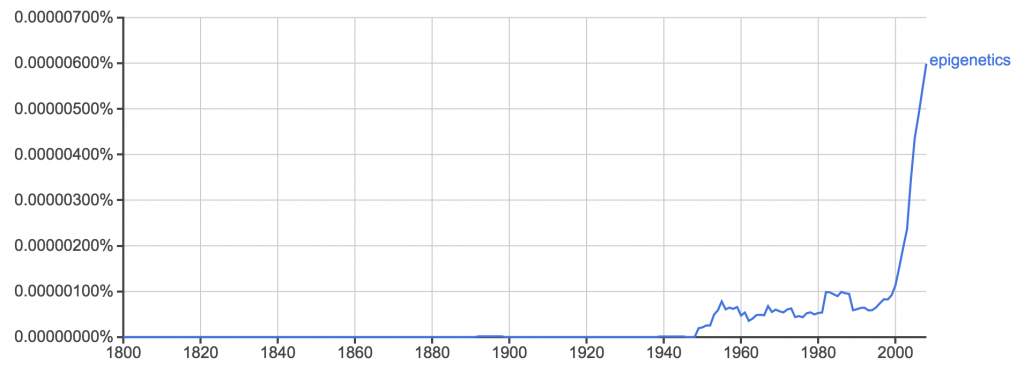
28 Comments